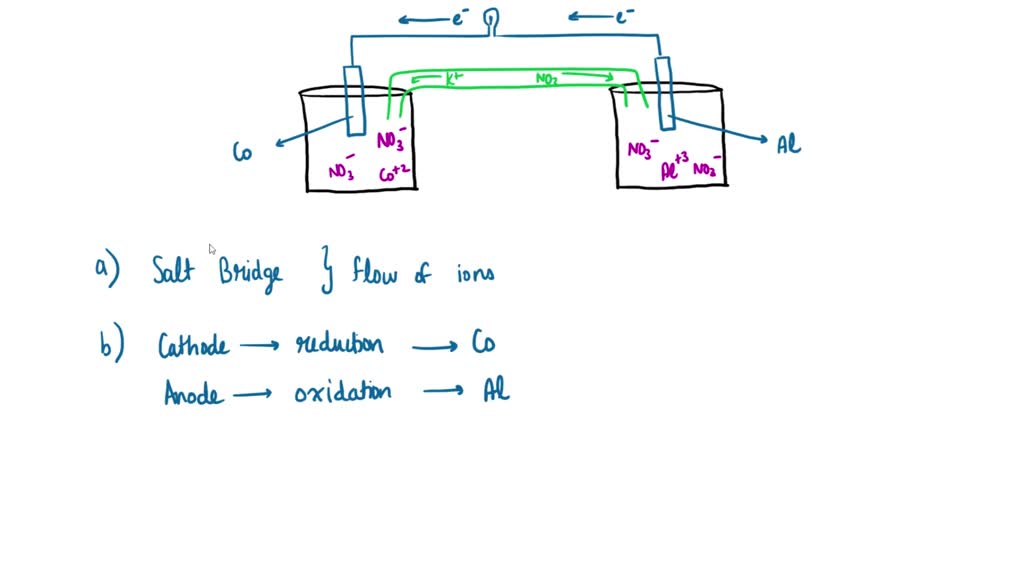
This flow of electrons is an electrical current that can be used to do work, such as turn a motor or power a light. The two electrodes must be electrically connected to each other, allowing for a flow of electrons that leave the metal of the anode and flow through this connection to the ions at the surface of the cathode. This forms a solid metal that deposits on the cathode. – At the cathode, the metal ion in the solution will accept one or more electrons from the cathode, and the ion’s oxidation state will reduce to 0. – The metal of the anode will oxidize, going from an oxidation state of 0 (in the solid form) to a positive oxidation state, and it will become an ion. In between these electrodes is the electrolyte, which contains ions that can freely move.Įlectron flow is observed at both the anode and cathode: the anode will undergo oxidation (loss of electrons) while the cathode experiences reduction (gain of electrons). Electrodes can be made from any sufficiently conductive materials, such as metals, semiconductors, graphite, and even conductive polymers. The zinc half-cell acts as the anode.Galvanic cells, also known as voltaic cells, are electrochemical cells in which spontaneous oxidation-reduction reactions produce electrical energy.Įlectrochemical cells have two conductive electrodes, called the anode and the cathode. Zinc is higher up on series than copper thus it is more easily oxidized.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
